 
The video below shows several different long chained oils, each progressively more viscous. Glycerol, CH 2OHCHOHCH 2OH, is viscous partly because of the length of the chain but also because of the extensive possibilities for hydrogen bonding between the molecules. Fuel oil, lubricating grease, and other long-chain alkane molecules are quite viscous for this reason. Imagine a styrofoam cup with a hole in the bottom. 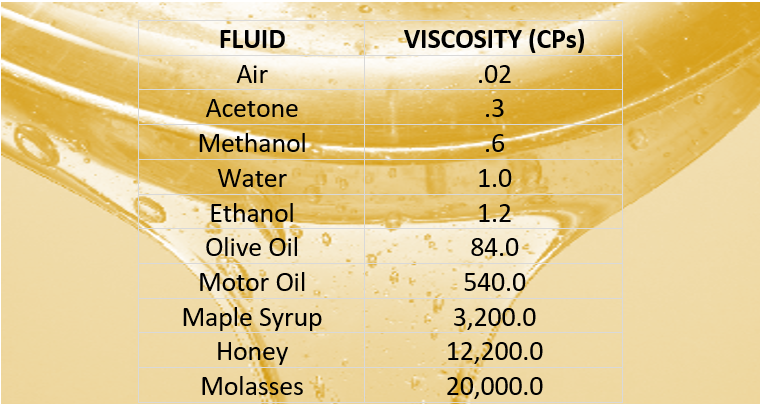
This is because the molecular chains get tangled up in each other like spaghetti-in order for the liquid to flow, the molecules must first unravel. What is viscosity This question is often best answered by example. Liquids containing long molecules are invariably very viscous. Honey, mostly glucose and fructose (see image below) is a good example of a liquid which owes its viscosity to hydrogen bonding. Liquids whose molecules are polar or can form hydrogen bonds are usually more viscous than similar nonpolar substances. Viscosity is governed by the strength of intermolecular forces and especially by the shapes of the molecules of a liquid. Those like ether or gasoline which flow very readily have low viscosities. Liquids which flow very slowly, like glycerin or honey, have high viscosities. The resistance to such flow is called the viscosity. Therefore, we’ve shown that corn syrup and honey have a higher viscosity (or are more viscous) than water and cooking oil.\)īecause its molecules can slide around each other, a liquid has the ability to flow. In our experiment, the marbles took longer to sink when dropped into the jars filled with corn syrup and honey than they did when dropped into the jars filled with water and cooking oil. The more viscous (or thick) a liquid is, the longer it will take for an object to move through the liquid. Viscosity can also be thought of as a measure of how “thick” a liquid is. Liquids that have a HIGH viscosity flow slowly (ie. water, rubbing alcohol, and vegetable oil). Liquids that have a LOW viscosity flow quickly (ie. Viscosity depends on the size and shape of the particles that make the liquid, as well as the attraction between the particles. Viscosity is a liquid’s resistance to flowing. The question answered in this experiment is: how does the consistency of a liquid impact how long it will take for a marble to sink in a jar of that liquid? A unique property of liquids is something called viscosity. Watch the Viscosity of Liquids Science Experiment Step by Step Instructions How Does the Science Experiment Work? Viscosity of Liquids Science Experiment Video Tutorial It also contains ideas on how you can expand on the experiment. Fluids with low viscosity have a low resistance and shear easily and the molecules flow quickly high viscosity fluids move sluggishly and resist deformation. 
Was your hypothesis correct? Do you know why some marbles sink to the bottom of the jar quickly and some marbles sink to the bottle of the jar slowly?įind out the answer in the how does this experiment work section below. You’ll notice right away that the marble behaves differently in each jar. Note: We have to remember that the viscosity depends upon the flowing capability of a fluid. 
The oil is all liquid substance and fat that is obtained after the processing of different seeds and fruits. Examples of substances that has high viscosity: Honey, Glues, Peanut butter, Long hydrocarbon chain, Ketchup and many more. Drop one marble at a time and observe what happens to the marble when it enters the liquid. List with 10 examples of viscous substances 1- Oil. Step 2 – Carefully drop one marble into each jar. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
